ニューロトランスミッター取り込みアッセイキット
ニューロトランスミッター取り込みアッセイキットとは
ニューロトランスミッター取り込みは、うつ病やアルツハイマー病、パーキンソン病などの神経変性疾患(NDD)において重要な役割を果たしている。セロトニン、ノルエピネフリン、ドーパミン・トランスポーター(それぞれSERT、NET、DAT)の活性を、トランスフェクタントあるいは初代発現細胞でモニターする能力は、これらの疾患の理解を深める鍵である。
ニューロトランスミッター取り込みアッセイキット
- ・ホモジニアス、ライブセル蛍光アッセイは、放射性アッセイに取って代わり、廃棄コストを削減し、放射性標識アッセイに関連する安全性リスクを排除する。
- ・ワンステップ、ホモジニアスアッセイフォーマットは、自動化とハイスループットスクリーニングに適している。
- ・2つのアッセイセットアップオプションにより、よりフレキシブルな対応が可能:ハイスループットなアプリケーションにはエンドポイントモード、メカニスティックな研究にはリアルタイムカイネティックモード。
最近まで、セロトニン、ノルエピネフリン、ドーパミントランスポーターの取り込みを測定するために、放射性標識化合物が使用されていた。ニューロトランスミッター取り込みアッセイキットの導入により、研究者はこれら3つの主要な神経伝達物質のライブセルでのカイネティック取り込みをスクリーニングするツールを手に入れた。
ニューロトランスミッター取り込みアッセイキットのデータ
-
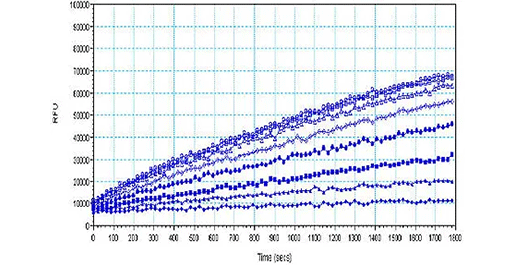
HEK-hSERT細胞におけるニソキセチン阻害作用
ヒト SERT を安定的に発現する HEK セルを、ポリ D-リジンでコートした 384 ウェルプレートに、1 ウェル当たり 10,000 細胞で O/N でプレーティングした。培地を除去し、HBSS/0.1% BSA 中のニソキセチン(公表 Ki = 383 nM)を、色素添加前に 37℃で 10 分間セルとインキュベートした。実験プロトコールに従ってアッセイを行い、モレキュラーデバイスのFlexStation® 3リーダーでカイネティック測定を30分間行った。 -
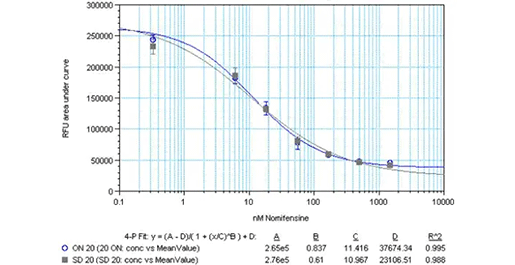
プレーティングの柔軟性
ヒトNETを安定に発現するHEK細胞を、ポリD-リジンコートした384ウェルプレートに、1ウェルあたり20,000細胞で一晩(青丸印)またはアッセイ当日に(黒四角印)プレーティングし、3時間接着させた。培地を除去し、HBSS/0.1% BSA中のノミフェンシン を、色素添加前に37℃で10分間セルとインキュベートした。アッセイをFlexStation® 3のカイネティック測定モードで30分間読み取った。阻害曲線は曲線下面積で表した。 -
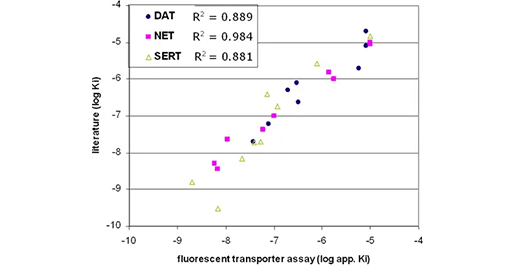
DAT、NET、SERT の IC50 相関(顧客結果)
ヒト DAT、NET、SERT を安定的に発現する細胞を 60,000 個/ウェル(96 ウエルプレート)で一晩プレーティングし、8 種類の既知の阻害剤で処理した。10 分後、色素溶液を添加し、上述のプロトコールに従ってアッセイを実施した。得られたデータからCheng-Prusoff式を用いてlog Kiを算出し、3つのトランスポーターすべてについて既知のKi値との相関を求めた。各トランスポーターの相関係数R2はグラフに示されており、全24値にわたる相関係数(R2値)は0.91であった。
ニューロトランスミッター取り込みアッセイキットの技術
-
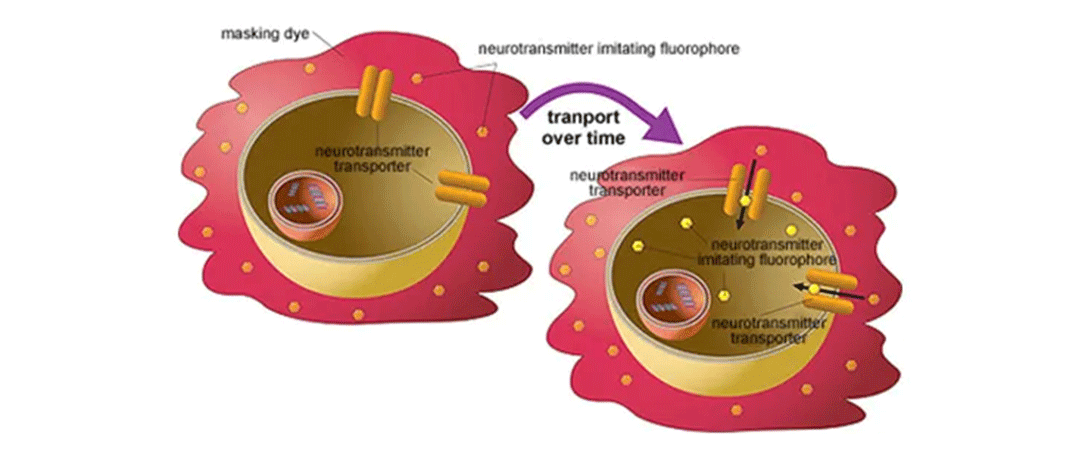
アッセイ原理-ニューロトランスミッター
ニューロトランスミッター取り込みアッセイキット(Neurotransmitter Transporter Uptake Assay Kit)は、生体アミン神経伝達物質を模倣した蛍光基材を使用し、それらの特異性トランスポーターを介して細胞内に取り込まれ、その結果、細胞内の蛍光強度が増加し、下方測定を行うマイクロプレートリーダーを用いてリアルタイムでモニターされる。この便利な洗浄不要の手順では、セルはキット試薬とインキュベートされ、評価のためにプレートリーダーに移される。このアッセイは、研究、アッセイ開発、スクリーニングのための学術、バイオテクノロジー、製薬環境での使用に理想的である。 -
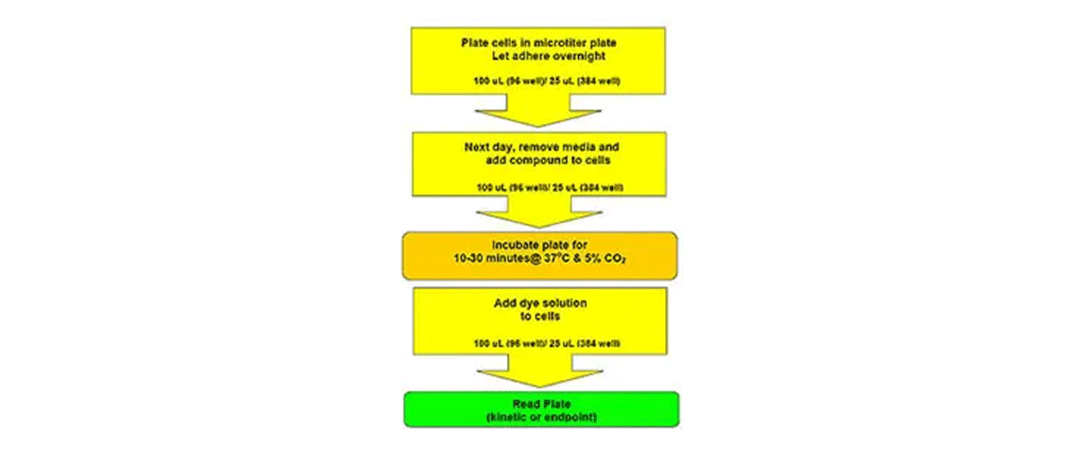
ワークフロー-ニューロトランスミッター
詳細については、モレキュラーデバイスのナレッジベースからアクセスできるニューロトランスミッター取り込みアッセイキットの製品添付文書を参照してください。
ニューロトランスミッター取り込みアッセイキットに対応する製品・サービス
-
SpectraMax iD3/iD5 マルチモードマイクロプレートリーダー

大型タッチスクリーンを備えた高感度マイクロプレートリーダー
-
Gemini XPSおよびEM マイクロプレートリーダー

フィルター不要の蛍光検出
-
FlexStation 3 マルチモードマイクロプレートリーダー

イオンチャネル・GPCR活性の測定に威力を発揮する
自動マルチチャンネルピペッター搭載マルチマイクロプレートリーダー -
SpectraMax Paradigm マルチモードマイクロプレートリーダー

高速で設定可能なマイクロプレートリーダー1台でハイスループットスクリーニングが可能
-
SpectraMax i3x マルチモードマイクロプレートリーダー

研究ニーズに合わせて進化できるマイクロプレートリーダー
-
FLIPR Penta ハイスループットセルベーススクリーニングシステム

リード化合物の同定および化合物の安全性評価を目的とした
ハイスループットカイネティックスクリーニングに理想的なシステム -
SpectraMax Mシリーズ マルチモードマイクロプレートリーダー

妥協ない性能と低価格を両立したマルチモードマイクロプレートリーダー
-
SpectraMax Mini マルチモードマイクロプレートリーダー

妥協ない性能と低価格を両立したマルチモードマイクロプレートリーダー